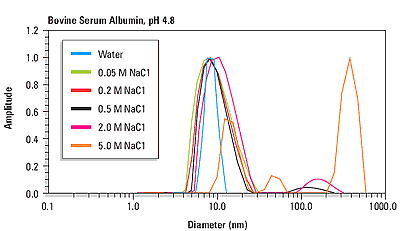
HT-DLS is a non-invasive, non-perturbative method of measuring protein and particle size distribution in terms of hydrodynamic radius (Rh) across a wide range, covering 0.5–1000 nm. 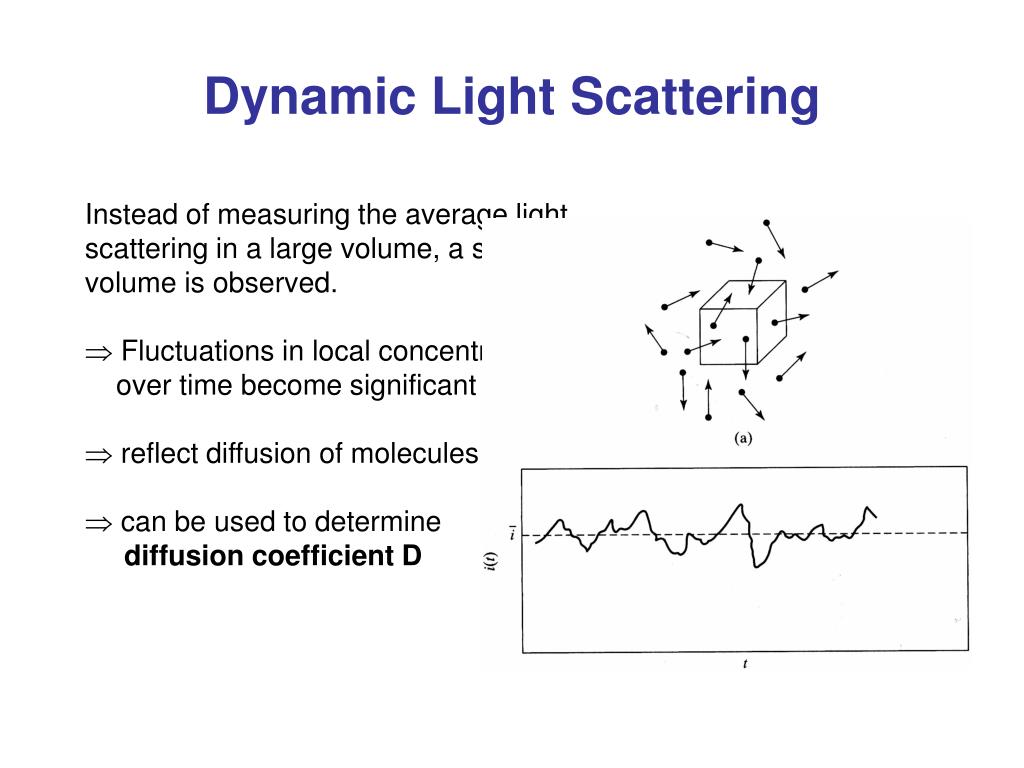
HT-DLS for Developability and Preformulation Studies Requiring just a few microlitres of solution for each sample in standard plates, unattended HT-DLS measures multiple biotherapeutic candidates in a variety of formulation and environmental conditions. A HT-DLS plate reader can be used as a standalone instrument or as part of a larger automated formulation workflow (2). HT-DLS performed in microwell plates typically provides 10–100× more measurements than would be collected using traditional cuvette-based DLS. In addition, viscosity can be measured with DLS using probe particles, such as common polystyrene latex beads.Īutomated, high-throughput dynamic light scattering (HT-DLS) helps streamline the candidate selection and preformulation processes. This makes DLS a particularly effective technique for assessing aggregation and stability in early, as well as late stages. The measurement takes place on timescales of seconds to minutes, and the sample is fully recoverable. DLS measures particle size and size distributions, from less than a nanometre and up to several micrometres, without disturbing the sample. One of the primary technologies for assessing aggregation, stability, and viscosity of biotherapeutics, from proteins, monoclonal antibodies, and peptides to attenuated viruses, is dynamic light scattering (DLS) (1). In a fully automated, microwell plate reader format, DLS constitutes a high-throughput biophysical screening technique for early-stage assessment of candidate developability and optimal formulation conditions.Īssessing a drug candidate’s suitability early in the discovery and development stages is essential for reducing the risk of costly downstream failure. Per sample, analytical column, 0.Daniel Some, Wyatt Technology Corp., Santa Barbara, California, USAĭynamic light scattering (DLS), widely recognized for its ability to detect aggregation in purified protein solutions, is actually far more versatile and may be applied to the evaluation of stability and viscosity of therapeutic proteins and other biopharmaceuticals. Per buffer, includes analytical column equilibration, BSA control, in addtion to per-sample fee. Per sample fee, in duplicate wells, 5-10 acquisitions/scan, 2 scans/well, in addition to plate setup fee. In addition to per-sample fee.ĭLS - sample data collection - macromolecules Setup fee per plate, up to 48 samples, includes BSA control and 1 buffer control. Required for samples with high molecular weight modifiers, such as glycosylation or detergent micelle will require additional analysisĭn/dc measurements of detergent or protein samples Hydrodynamic Radius from DLS (when light scattering signal is sufficient) Includes:Ĭolumn equilibration with your buffer (pH at or below pH 7.5) or with PBSīSA Standard to test column performance and normalize detectors Prior purification by size-exclusion chromatography (SEC) is strongly recommended. Size-exclusion Chromatography with Multi-Angle Light Scattering (SEC-MALS)ĬMI SEC-MALS System: Wyatt Dawn Heleos with in-line DLS, RI UV, Agilent chromatography Standard SEC-MALS Serviceįor proteins and protein complexes between 15 KDa and 1 MDa, without high molecular weight modifiers (modifiers of mass >5% total).
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
